Piroplasmosis
|
Summary
Equine Piroplasmosis (EP) is a blood borne disease carried by ticks that in the acute form can cause hemolytic anaemia, fever, weight loss, jaundice, colic, diarrhoea, liver damage, abortion and even death. It is present throughout France. Acute disease mostly occurs in naive horses that have moved from an area without EP (e.g. the UK) to an area where EP is endemic (like France). Diagnosis is now mostly by blood slide and PCR. Treatment for infected horses is 2 to 4 intra-muscular injections of Imidocarb (called Carbesia in France), depending on whether the aim of treatment is to eliminate just the clinical signs of disease or the parasitic infection (which may not be desirable in areas where EP is endemic), plus good nursing, diet, and supportive care in severe cases. Iron should not be supplemented to horses with piroplasmosis. Horses gain life-long immunity following infection (as long as low levels of infection remain). The best way to prevent piroplasmosis is to prevent ticks feeding on a horse, by checking horses every day for ticks and removing ticks immediately when found (a Tire-Tique is good for removing ticks). and/or using an insect repellent, and where possible avoiding long grass/plants and bushes. |
What is Equine Piroplasmosis (EP)?
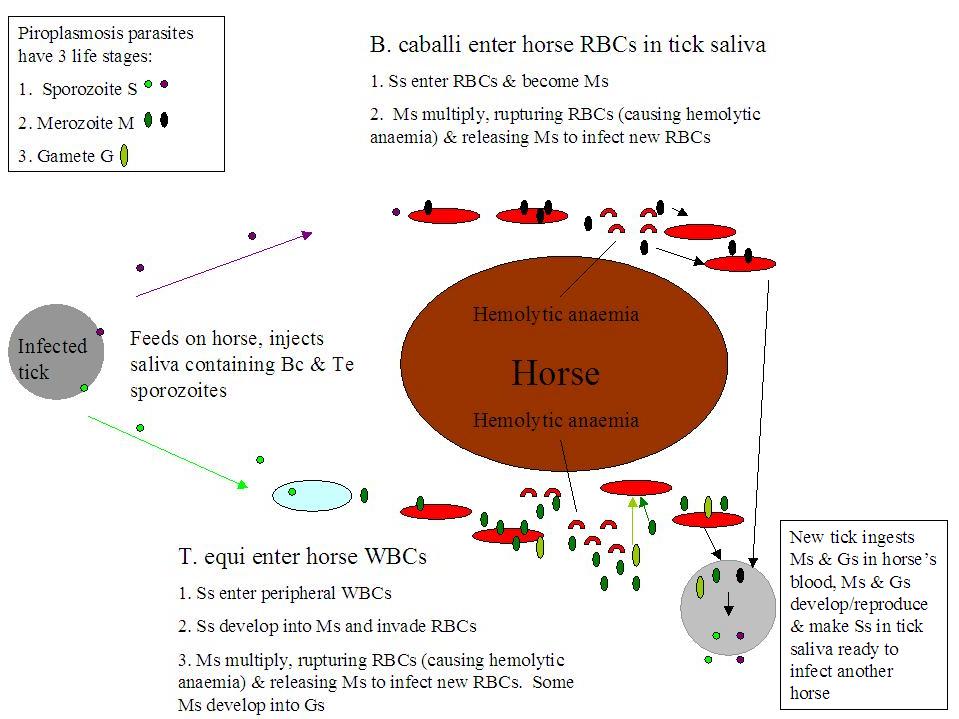
Equine piroplasmosis describes infection by either or both of 2 protozoan (single-celled animal) parasites: Babesia caballi (B. caballi) and/or Theileria equi (T. equi). EP is sometimes called Babesiosis (infection by B. caballi alone) or Theileriosis (infection by T. equi alone), but the clinical signs of both diseases are so similar that equine piroplasmosis has become the blanket term to cover both infections. Generally infection with T. equi causes a more severe clinical disease. Feeding ticks pick up the parasites from an infected horse and inject them in their saliva into an uninfected ("naive") horse. The parasites can also be spread by contaminated blood, e.g. on needles, surgical instruments/dental tools or by blood transfusion. Once a tick is infected with the parasite, it can pass the infection to its offspring, so generations of ticks can transmit the disease without having to find another infected host (B. caballi only). Once inside the horse, the parasites invade the red blood cells (RBCs), where they multiply, causing the infected RBCs to rupture and more RBCs to be infected. In addition, RBCs are identified as contaminated and destroyed as they pass through the spleen, and the spleen destroys some uninfected RBCs too. This destruction of RBCs causes hemolytic anaemia (anaemia is described as hemolytic when RBCs are destroyed earlier than normal).
Clinical signs
Some horses become infected with equine piroplasmosis without showing any clinical signs (asymptomatic carriers), others show acute or chronic signs of the disease. The acute form can be mild and pass quickly, or can cause severe illness. It takes 10 to 30 days for a horse to show signs of acute piroplasmosis after being infected by a tick.
Signs of acute piroplasmosis often start with:
- Fever/above normal temperature (often exceeds 40'C). The horse's normal temperature is 37.5 to 38.5'C (although it has recently been found that individual differences mean that you should know your horse's normal temperature). For how to take the temperature see: How to take a horse's temperature - BHS. Note that it is worth spending a bit of extra money on a thermometer that beeps when ready, and that only takes 5 to 10 seconds.
- Going off feed/inappetence/anorexia, leading to weight loss.
- Lethargy.
- General malaise/being "not quite right".
- Increased heart rate (should be 28 to 44 beats per minute) and respiration rate (should be 8 to 16 breaths per minute). For how to take the heart rate and respiration rate, see: How to take a horse's pulse and respiratory rate - BHS (the video shows how to use both a stethoscope and the mandibular pulse for the heart rate. Remember you can also use the digital pulse in the fetlock or pastern).
- Mild colic (which may be followed by diarrhoea)
- Constipation (small dry dung or reduced passing of dung) (which may be followed by diarrhoea)
- Diarrhoea
- Peripheral oedema - swelling, particularly of the legs and/or below the belly
- Weakness in the hind end/swaying/ataxia, possibly seizures
- An enlarged spleen (this may be felt on rectal examination).
- Hemolytic anaemia - (the form of anaemia cased when red blood cells are destroyed earlier than they should be - in piroplasmosis the parasites cause RBCs to rupture and the spleen destroys infected RBCs). Hemolytic anaemia causes:
- Weakness,
- Blood in the urine (hemoglobulinuria - blood looks darker than normal/reddish/brown),
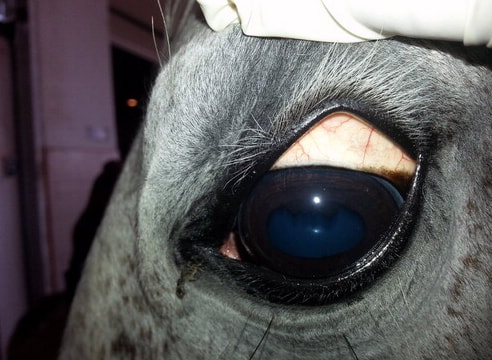
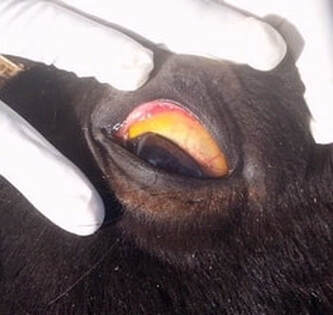
- Jaundice (technically called icterus) - yellow colour to the mucous membranes (eyelids, gums) (mucous membranes may also be pale due to anaemia),
- Petechiae (tiny red/purple dots) may be seen on the mucous membranes (gums, eyelids) (due to thrombocytopenia or low platelets).
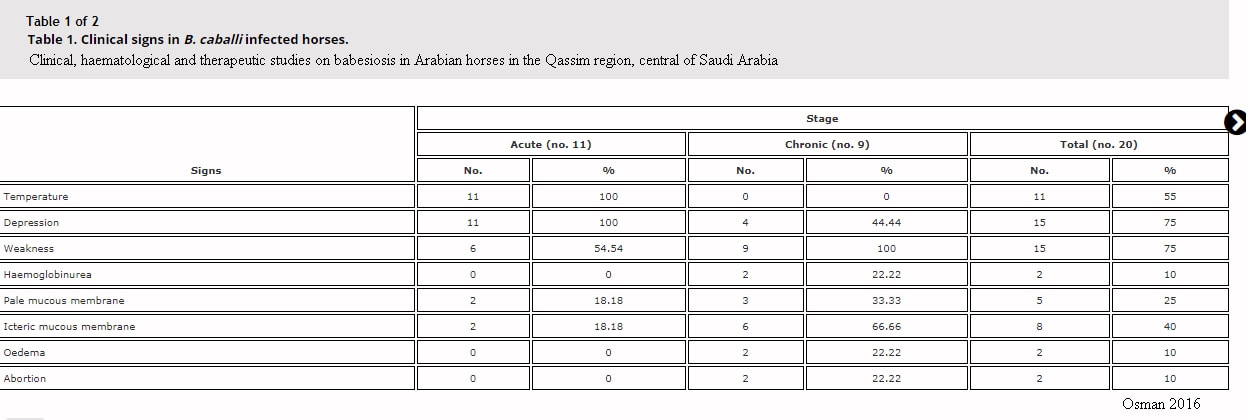
The images above and table below come from the Open Access paper:
Osman SA
Clinical, haematological and therapeutic studies on babesiosis in Arabian horses in the Qassim region, central of Saudi Arabia
Journal of Applied Animal Research 2017, 45:1, 118-121, DOI: 10.1080/09712119.2015.1124339
Osman SA
Clinical, haematological and therapeutic studies on babesiosis in Arabian horses in the Qassim region, central of Saudi Arabia
Journal of Applied Animal Research 2017, 45:1, 118-121, DOI: 10.1080/09712119.2015.1124339
In more severe cases complications can include
Signs of chronic piroplasmosis are usually non-specific and include weakness, lethargy, poor performance/exercise intolerance, loss of appetite, weight loss, loss of condition, poor hair coat, transient fever and hemolytic anaemia. The spleen may be enlarged on rectal examination.
- liver failure, kidney damage/acute renal failure, secondary infections e.g. pneumonia, and systemic inflammatory response syndrome.
- Pregnant mares can pass piroplasmosis to the foetus, causing abortion and birth of stillborn foals. Infected live foals may develop clinical signs quickly, including anaemia and jaundice, or not show clinical signs and become healthy carriers.
Signs of chronic piroplasmosis are usually non-specific and include weakness, lethargy, poor performance/exercise intolerance, loss of appetite, weight loss, loss of condition, poor hair coat, transient fever and hemolytic anaemia. The spleen may be enlarged on rectal examination.
Diagnosis
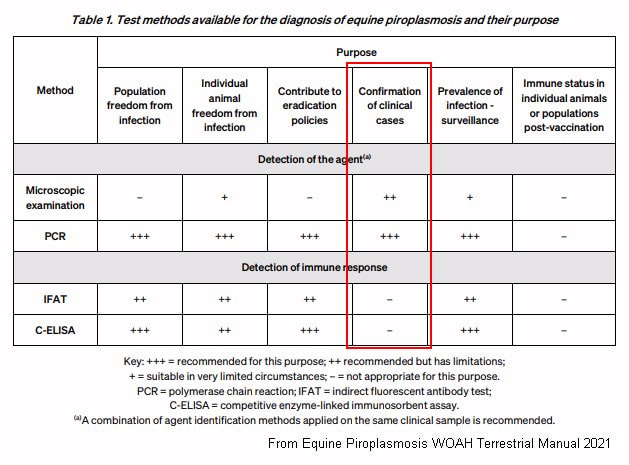
Diagnosis of equine piroplasmosis consists of specifically looking for parasitic infection - blood smear under the microscope and/or sending off blood for PCR and/or serological testing, supported by CBC (complete blood count) and biochemistry findings (CBC and biochemistry will usually be run by your vet so results should be available quickly). Normalization of the CBC may be a good indicator of response to treatment, and biochemistry should be monitored until liver and kidney enzymes are normal.
Complete Blood Count (CBC)
Anaemia is diagnosed by below normal red blood cell count (GR/hematies), haemoglobin (Hb/hemoglobine) and PCV/hematocrit (Hte/hematocrite) (in brackets are names that you might see on French blood test results). PCV can be as low as 10% but rarely decreases below 20%.
The anaemia is initially normocytic (MCV is normal), then becomes macrocytic (MCV is above normal).
MCH and MCHC are variable (will generally increase but may decrease)
Thrombocytopenia (decreased platelets) is often seen.
White blood cells and fibrinogen vary depending on the stage of infection and severity.
Diagnosis of equine piroplasmosis consists of specifically looking for parasitic infection - blood smear under the microscope and/or sending off blood for PCR and/or serological testing, supported by CBC (complete blood count) and biochemistry findings (CBC and biochemistry will usually be run by your vet so results should be available quickly). Normalization of the CBC may be a good indicator of response to treatment, and biochemistry should be monitored until liver and kidney enzymes are normal.
Complete Blood Count (CBC)
Anaemia is diagnosed by below normal red blood cell count (GR/hematies), haemoglobin (Hb/hemoglobine) and PCV/hematocrit (Hte/hematocrite) (in brackets are names that you might see on French blood test results). PCV can be as low as 10% but rarely decreases below 20%.
The anaemia is initially normocytic (MCV is normal), then becomes macrocytic (MCV is above normal).
MCH and MCHC are variable (will generally increase but may decrease)
Thrombocytopenia (decreased platelets) is often seen.
White blood cells and fibrinogen vary depending on the stage of infection and severity.
Biochemistry
Biochemistry changes may include:
decreases in: total protein, perhaps also albumin
increases in: bilibrubin, liver enzymes (AST, GGT, ALP, perhaps also LDH), CK and perhaps also urea. (Increased bilirubin can be an indicator of hemolytic anaemia. Raised liver enzymes may be because of reduced blood flow to the liver).
Biochemistry changes may include:
decreases in: total protein, perhaps also albumin
increases in: bilibrubin, liver enzymes (AST, GGT, ALP, perhaps also LDH), CK and perhaps also urea. (Increased bilirubin can be an indicator of hemolytic anaemia. Raised liver enzymes may be because of reduced blood flow to the liver).
Onyiche TE, Suganuma K, Igarashi I, Yokoyama N, Xuan X, Thekisoe O.
A Review on Equine Piroplasmosis: Epidemiology, Vector Ecology, Risk Factors, Host Immunity, Diagnosis and Control.
Int J Environ Res Public Health. 2019 May 16;16(10):1736. doi: 10.3390/ijerph16101736. PMID: 31100920; PMCID: PMC6572709.
A Review on Equine Piroplasmosis: Epidemiology, Vector Ecology, Risk Factors, Host Immunity, Diagnosis and Control.
Int J Environ Res Public Health. 2019 May 16;16(10):1736. doi: 10.3390/ijerph16101736. PMID: 31100920; PMCID: PMC6572709.
Microscopy - blood smear
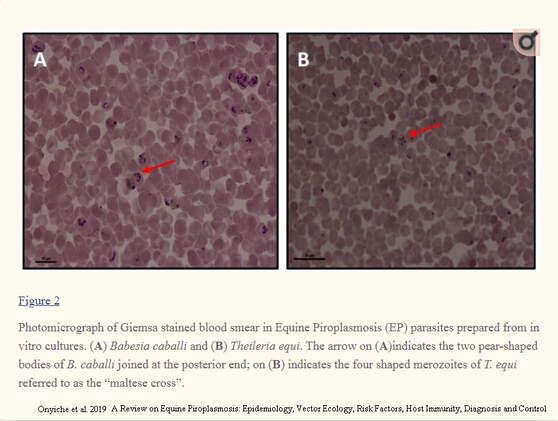
During the acute phase a blood smear (initially thin, but thick may increase sensitivity if examined by someone experienced) should be made from superficial capillary blood (e.g. from the ear) and examined under the microscope for the presence of the parasites. Seeing (and accurately identifying) the parasites is diagnostic for piroplasmosis (see image above left), but not seeing the parasites does not rule out piroplasmosis as the level of parasites is low even in the acute phase* (particularly for B. caballi infections). Microscopy is unlikely to be useful for chronic cases.
*T. equi usually infects 1-7% of RBCs in horses showing signs of clinical disease, but may infect >20% (and up to 95%).
B. caballi rarely infects more than 1% of RBCs and may infect <0.1%.
During the acute phase a blood smear (initially thin, but thick may increase sensitivity if examined by someone experienced) should be made from superficial capillary blood (e.g. from the ear) and examined under the microscope for the presence of the parasites. Seeing (and accurately identifying) the parasites is diagnostic for piroplasmosis (see image above left), but not seeing the parasites does not rule out piroplasmosis as the level of parasites is low even in the acute phase* (particularly for B. caballi infections). Microscopy is unlikely to be useful for chronic cases.
*T. equi usually infects 1-7% of RBCs in horses showing signs of clinical disease, but may infect >20% (and up to 95%).
B. caballi rarely infects more than 1% of RBCs and may infect <0.1%.
|
PCR/Serology
To confirm that a horse has current infection with piroplasmosis, PCR testing should be carried out. Same day PCR testing is carried out at Equisyn Equine Vet Clinic near 16260 Chasseneuil-sur-Bonnieure. The PCR profile run is the Piro-Like/tick-borne disease profile also run by Frank Duncombe/Labeo, supported by Labeo, and includes both forms of piroplasmosis, anaplasmosis, Lyme disease and leptospirosis. PCR testing is available to non-clients as well as clients of the clinic. Other laboratories in France that test for piroplasmosis include Frank Duncombe/Labeo and Vetodiag. PCR (polymerase chain reaction) looks for and confirms the presence of parasite DNA and is considered highly specific and sensitive. PCR can detect low levels of parasitic infection and can be used for chronic infections. |
Serology measures the horse's immune response (the presence of antibodies) to the parasitic infection, and can detect carriers without symptoms as well as clinical infections, but cannot distinguish between whether the horse is currently infected or has been infected. Horses infected with T. equi produce antibodies against merozoites within 7 to 11 days, with levels peaking at 30 to 45 days. Horses infected with B. caballi also produce antibodies but less is known about this response. Serological tests include IFAT, ELISA and Western blot. None of these tests are perfect - a highly positive result is likely to be positive (although false positives are possible) but a negative result may not rule out infection - and ideally at least 2 different tests should be done.
Treatment
Treatment is the antiprotozoal drug Imidocarb, which is called Carbesia in France, given by intramuscular injection. In countries where equine piroplasmosis is endemic, treatment is usually aimed at eliminating clinical signs of the disease without eliminating the parasite from the body, as immunity depends on continued low level presence of the parasite, but where infected horses have been brought into a piroplasmosis-free country, the aim of treatment will be parasite elimination. T. equi is more difficult to treat than B. caballi. Recommended treatment for elimination of the parasites suggests 2 doses of 2.2 mg/kg 24 hours apart for B. caballi, and 4 doses of 4 mg/kg 72 hours apart for T. equi - treatment is likely to be less than this in countries where EP is endemic (e.g. France).
Imidocarb/Carbesia is an anticholinesterase and treated horses should be monitored for associated side effects such as colic, diarrhoea, increased salivation, sweating, heavy breathing, agitation and recumbency, and the vet will usually give injections of NSAIDs (anti-inflammatory pain relief) e.g. Nefotek (ketoprofen) and/or anti-spasmodic e.g. Buscopan, at the same time. Side effects are usually seen immediately after Imidocarb has been given and are rarely life-threatening. Swelling and muscle inflammation around the injection site are common, and injection sites should be rotated and monitored. Imidocarb is cleared by the liver and kidneys and at high doses toxicity can damage these organs. Note that donkeys and mules are particularly sensitive to Imidocarb and should not receive the higher dose.
Treatment should include supportive care for clinical signs as appropriate, and could involve anti-inflammatory medication (NSAIDS), pain relief, fluid treatment (e.g. intravenous fluid) for dehydrated horses/horses with kidney damage, and rarely, blood transfusion for cases with severe anaemia.
Management and Diet
Management includes appropriate nursing, rest and a good diet that provides recommended levels of energy, protein, minerals, vitamins and essential fatty acids - but not increased iron. Vets sometimes suggest an iron or "blood building" supplement (e.g. Red Cell) for horses with piroplasmosis. Such supplements usually contain large amounts of iron and are not recommended for horses with hemolytic anaemia (or any form of anaemia other than possibly severe haemorrhage and blood loss). With hemolytic anaemia, red blood cells are broken down in the body, releasing iron, which is locked away by the body until it is recycled into new red blood cells. Iron is toxic and difficult to get rid of from the body. Besides, the horse's natural forage diet is high in iron. See Anaemia and Iron Supplements.
Supporting immunity and new RBC development is important, and specifically the diet should include quality protein, e.g. micronized whole soya beans or whey protein/whey protein isolate, and provide at least 100% and probably 150-200% of the RDA for copper (important for RBC building and almost always deficient in a forage diet), the RDA being 100 mg for a 500 kg horse, so 150 to 200 mg/day copper - a horse eating 2% bodyweight in grass or hay is likely to ingest 40 to 60 mg of copper, leaving 90 to 160 mg copper to be supplied in bucket feed. Copper should be balanced by zinc in a 1 copper to 4 zinc ratio, so if providing 150 mg copper in the total diet, provide 600 mg zinc in the total diet. Plus 1 to 1.5 mg selenium in the total diet. If the horse is not eating well, give appetizing, easy to digest feed, ideally based on rapidly fermentable fibre such as sugar beet to provide slow release energy and to carry minerals and protein. Try to avoid making sudden significant changes to the diet. Grass is likely to be good for horses recovering from piroplasmosis, but be extra cautious with horses with Equine Metabolic Syndrome/insulin dysregulation, as piroplasmosis is a systemic inflammatory disease which could increase insulin dysregulation and therefore the risk of laminitis.
Immunity
Acute disease is uncommon in areas where piroplasmosis is endemic. Once a horse has been infected with either B. caballi or T. equi, it develops immunity against that form of disease, and (if not treated until elimination of the parasites) becomes a carrier. Without treatment, horses infected with B. caballi may completely eliminate it from their blood after around 3 to 5 years, but horses infected with T. equi remain life-long carriers. If brood mares are infected, foals can be infected in the womb and be healthy carriers. New born foals may be exposed to the parasites while still protected by maternal antibodies.
To prevent / help identify piroplasmosis
Websites with information about equine piroplasmosis/Research papers
Equine Piroplasmosis chapter 3.6.8. WOAH Terrestrial Manual 2021
Int J Environ Res Public Health. 2019 May; 16(10): 1736. doi: 10.3390/ijerph16101736
A Review on Equine Piroplasmosis: Epidemiology, Vector Ecology, Risk Factors, Host Immunity, Diagnosis and Control
Onyiche et al.
Equine Piroplasmosis - OIE Terrestrial Manual 2018
Equine Piroplasmosis - Anna Spickler, 2018
FAQ Regarding Equine Piroplasmosis (EP) - AAEP
Equine piroplasmosis: What, how and why - Ed Kane 2016 - DVM360
Adam MI, Pikalo J, Snyder A, Steinrigl A, Koeller G, Schusser GF
Equine Piroplasmosis – a case of severe Babesia caballi infection associated with acute renal failure
Berliner und Münchener tierärztliche Wochenschrift Nov 2016 130:113 - 118. DOI: 10.2376/0005-9366-16064
(full pdf available online)
Osman SA
Clinical, haematological and therapeutic studies on babesiosis in Arabian horses in the Qassim region, central of Saudi Arabia
Journal of Applied Animal Research 2017, 45:1, 118-121, DOI: 10.1080/09712119.2015.1124339
Scoles GA, Ueti MW
Vector Ecology of Equine Piroplasmosis
Annual Review of Entomology Jan 2015 Vol 60: 561-580 https://doi.org/10.1146/annurev-ento-010814-021110
Wise LN, Pelzel-McCluskey AM, Mealey RH, Knowles DP
Equine Piroplasmosis
Vet Clin North Am Equine Pract. 2014 Dec;30(3):677-93. doi: 10.1016/j.cveq.2014.08.008 (DeepDyve)
J Vet Intern Med 2013;27:1334–1346
Review of Equine Piroplasmosis
L.N. Wise, L.S. Kappmeyer, R.H. Mealey, and D.P. Knowles
Equine Piroplasmosis - USDA APHIS 2020
APHIS Factsheet: Equine Piroplasmosis - USDA APHIS 2009
A Literature Review of Equine Piroplasmosis - USDA APHIS 2010
Traub-Dargatz JL, Short MA, Pelzel AM,Norman TE, Knowles DP
Equine Piroplasmosis
AAEP Proceedings Vol 56 2010
Equine Piroplasmosis - OIE World Animal Health 2009
Equine Piroplasmosis in Equine Infectious Diseases - edited by Debra C. Sellon, Maureen T. Long 2007
About Equine Piroplasmosis - Veterinary Diagnostic Services, University of Glasgow
Information in French
Piroplasmoses - RESPE
Rakover, Pauline PhD thesis November 2018
La Piroplasmose: Parasites, Vecteurs et Traitements
Carbesia (Imidocarb) RCP (datasheet)
La piroplasmose chez le cheval : une maladie parasitaire du système sanguin - www.classequine.com
Treatment
Treatment is the antiprotozoal drug Imidocarb, which is called Carbesia in France, given by intramuscular injection. In countries where equine piroplasmosis is endemic, treatment is usually aimed at eliminating clinical signs of the disease without eliminating the parasite from the body, as immunity depends on continued low level presence of the parasite, but where infected horses have been brought into a piroplasmosis-free country, the aim of treatment will be parasite elimination. T. equi is more difficult to treat than B. caballi. Recommended treatment for elimination of the parasites suggests 2 doses of 2.2 mg/kg 24 hours apart for B. caballi, and 4 doses of 4 mg/kg 72 hours apart for T. equi - treatment is likely to be less than this in countries where EP is endemic (e.g. France).
Imidocarb/Carbesia is an anticholinesterase and treated horses should be monitored for associated side effects such as colic, diarrhoea, increased salivation, sweating, heavy breathing, agitation and recumbency, and the vet will usually give injections of NSAIDs (anti-inflammatory pain relief) e.g. Nefotek (ketoprofen) and/or anti-spasmodic e.g. Buscopan, at the same time. Side effects are usually seen immediately after Imidocarb has been given and are rarely life-threatening. Swelling and muscle inflammation around the injection site are common, and injection sites should be rotated and monitored. Imidocarb is cleared by the liver and kidneys and at high doses toxicity can damage these organs. Note that donkeys and mules are particularly sensitive to Imidocarb and should not receive the higher dose.
Treatment should include supportive care for clinical signs as appropriate, and could involve anti-inflammatory medication (NSAIDS), pain relief, fluid treatment (e.g. intravenous fluid) for dehydrated horses/horses with kidney damage, and rarely, blood transfusion for cases with severe anaemia.
Management and Diet
Management includes appropriate nursing, rest and a good diet that provides recommended levels of energy, protein, minerals, vitamins and essential fatty acids - but not increased iron. Vets sometimes suggest an iron or "blood building" supplement (e.g. Red Cell) for horses with piroplasmosis. Such supplements usually contain large amounts of iron and are not recommended for horses with hemolytic anaemia (or any form of anaemia other than possibly severe haemorrhage and blood loss). With hemolytic anaemia, red blood cells are broken down in the body, releasing iron, which is locked away by the body until it is recycled into new red blood cells. Iron is toxic and difficult to get rid of from the body. Besides, the horse's natural forage diet is high in iron. See Anaemia and Iron Supplements.
Supporting immunity and new RBC development is important, and specifically the diet should include quality protein, e.g. micronized whole soya beans or whey protein/whey protein isolate, and provide at least 100% and probably 150-200% of the RDA for copper (important for RBC building and almost always deficient in a forage diet), the RDA being 100 mg for a 500 kg horse, so 150 to 200 mg/day copper - a horse eating 2% bodyweight in grass or hay is likely to ingest 40 to 60 mg of copper, leaving 90 to 160 mg copper to be supplied in bucket feed. Copper should be balanced by zinc in a 1 copper to 4 zinc ratio, so if providing 150 mg copper in the total diet, provide 600 mg zinc in the total diet. Plus 1 to 1.5 mg selenium in the total diet. If the horse is not eating well, give appetizing, easy to digest feed, ideally based on rapidly fermentable fibre such as sugar beet to provide slow release energy and to carry minerals and protein. Try to avoid making sudden significant changes to the diet. Grass is likely to be good for horses recovering from piroplasmosis, but be extra cautious with horses with Equine Metabolic Syndrome/insulin dysregulation, as piroplasmosis is a systemic inflammatory disease which could increase insulin dysregulation and therefore the risk of laminitis.
Immunity
Acute disease is uncommon in areas where piroplasmosis is endemic. Once a horse has been infected with either B. caballi or T. equi, it develops immunity against that form of disease, and (if not treated until elimination of the parasites) becomes a carrier. Without treatment, horses infected with B. caballi may completely eliminate it from their blood after around 3 to 5 years, but horses infected with T. equi remain life-long carriers. If brood mares are infected, foals can be infected in the womb and be healthy carriers. New born foals may be exposed to the parasites while still protected by maternal antibodies.
To prevent / help identify piroplasmosis
- There is no vaccine for equine piroplasmosis.
- Check for ticks every day, particularly: around and inside the ears, under the forelock and mane, under the jaw, around the girth & between the front legs, and all down the legs, particularly the back of the fetlock and pastern, around the top of the hind legs/thighs and sheath/udder area, right down the hind legs, particulary the back of the fetlock and pastern, and the length of the dock. Ticks like thin skin and often hide under hair and in skin folds.
- Remove ticks if found, being careful not to leave any part of the tick in the horse - a Tire-Tique which uses a twisting action is good for removing ticks whole. Not removing a tick correctly can stimulate the tick to secrete more saliva, cause regurgitation of ingested blood, introduce other tick-body secretions or excretions into the wound, or increase the risk of secondary infection around the bite. After removing a tick wash the area and/or apply antiseptic
- Treat horses with an insect repellent that deters ticks, usually with the active ingredient pyrethrin, pyrethroid or permethrin, e.g. diluted Butox (active ingredient deltramethrin, a pyrethroid), Coopers fly repellent (active ingredient permethrin).
- Fly rugs and leg wraps may help deter ticks from climbing onto horses (author's theory).
- Ticks climb to the tips of grasses and shrubs (but not trees) to wait for host animals to brush by, then crawl onto the host, so try to keep horses away from long grass, plants and bushes. Top/mow fields and cut back borders (but without harming wildlife like hedgehogs!) and check horses carefully after hacking through long grass/plants.
- Risk increases with immunological naivety (i.e. horses that haven't previously been exposed to EP - be cautious about keeping horses from the UK with or near horses born in France or other countries where EP is endemic) and with increased density of infected ticks and infected horses. Horses suffering from stress, a low immune response or with their spleen removed may be more susceptible to/at greater risk from EP. Carriers have had relapses associated with stress, strenuous exercise, immunosuppression and steroid administration.
- Ensure the every day diet meets requirements for protein, minerals, vitamins and essential fatty acids, and particularly copper (which is important for red blood cell development), and zinc (to balance copper) and selenium - these 3 minerals are almost always deficient in a forage diet. Ideally feed a balancer that supplies 100 mg copper, 400 mg zinc and 1 mg selenium to a 500 kg horse. Do not supplement iron (other than low levels found in some balancers/feeds (or unless a horse is not eating good amounts of forage when a balancer/mineral supplement supplying reasonable levels of iron may be fed) - the RDA of 400 mg for a 500 kg horse is easily met for most horses eating 1.5-2% bodyweight in grass, hay or haylage). See Diet.
- Do NOT feed garlic - garlic has been shown to cause hemolytic anaemia at 16 g/day for a 500 kg horse.
- Check your horse's temperature, heart rate and respiration rate at the first sign he/she may not be quite right.
- Watch your horse urinating and check the urine is light creamy/yellowy, not red or brown (however this is not an early sign of piroplasmosis and not always seen).
Websites with information about equine piroplasmosis/Research papers
Equine Piroplasmosis chapter 3.6.8. WOAH Terrestrial Manual 2021
Int J Environ Res Public Health. 2019 May; 16(10): 1736. doi: 10.3390/ijerph16101736
A Review on Equine Piroplasmosis: Epidemiology, Vector Ecology, Risk Factors, Host Immunity, Diagnosis and Control
Onyiche et al.
Equine Piroplasmosis - OIE Terrestrial Manual 2018
Equine Piroplasmosis - Anna Spickler, 2018
FAQ Regarding Equine Piroplasmosis (EP) - AAEP
Equine piroplasmosis: What, how and why - Ed Kane 2016 - DVM360
Adam MI, Pikalo J, Snyder A, Steinrigl A, Koeller G, Schusser GF
Equine Piroplasmosis – a case of severe Babesia caballi infection associated with acute renal failure
Berliner und Münchener tierärztliche Wochenschrift Nov 2016 130:113 - 118. DOI: 10.2376/0005-9366-16064
(full pdf available online)
Osman SA
Clinical, haematological and therapeutic studies on babesiosis in Arabian horses in the Qassim region, central of Saudi Arabia
Journal of Applied Animal Research 2017, 45:1, 118-121, DOI: 10.1080/09712119.2015.1124339
Scoles GA, Ueti MW
Vector Ecology of Equine Piroplasmosis
Annual Review of Entomology Jan 2015 Vol 60: 561-580 https://doi.org/10.1146/annurev-ento-010814-021110
Wise LN, Pelzel-McCluskey AM, Mealey RH, Knowles DP
Equine Piroplasmosis
Vet Clin North Am Equine Pract. 2014 Dec;30(3):677-93. doi: 10.1016/j.cveq.2014.08.008 (DeepDyve)
J Vet Intern Med 2013;27:1334–1346
Review of Equine Piroplasmosis
L.N. Wise, L.S. Kappmeyer, R.H. Mealey, and D.P. Knowles
Equine Piroplasmosis - USDA APHIS 2020
APHIS Factsheet: Equine Piroplasmosis - USDA APHIS 2009
A Literature Review of Equine Piroplasmosis - USDA APHIS 2010
Traub-Dargatz JL, Short MA, Pelzel AM,Norman TE, Knowles DP
Equine Piroplasmosis
AAEP Proceedings Vol 56 2010
Equine Piroplasmosis - OIE World Animal Health 2009
Equine Piroplasmosis in Equine Infectious Diseases - edited by Debra C. Sellon, Maureen T. Long 2007
About Equine Piroplasmosis - Veterinary Diagnostic Services, University of Glasgow
Information in French
Piroplasmoses - RESPE
Rakover, Pauline PhD thesis November 2018
La Piroplasmose: Parasites, Vecteurs et Traitements
Carbesia (Imidocarb) RCP (datasheet)
La piroplasmose chez le cheval : une maladie parasitaire du système sanguin - www.classequine.com
Frequently Asked Questions
Q. How long must a tick be on a horse before piroplasmosis is transmitted?
A. This will depend on how quickly the tick's saliva passes into the horse, and how quickly the parasite sporozoites are released into the saliva.
The RESPE article Piroplasmoses suggests that transmission of the piroplasmosis parasites happens a few days after the tick becomes attached, because the parasites have to go through a development phase, and says that ticks should be removed as soon as they are seen. Sporozoites are the form of the parasite that pass from the tick to the horse, and they generally start to mature in the tick's salivary glands ready for transmission to the horse only once the tick attaches itself to the horse and starts to feed on blood. Once mature, sporozoites pass into the horse's blood in the tick's saliva as the tick feeds. However, in other Theileria species, sporozoites have been found to mature before the tick attaches for feeding, when temperatures are high, so it may be that T. equi can pass to a horse within hours of a tick attaching in hot weather (however the ticks in the CHG area of France tend to not be active in hot weather). Jalovecka 2018 states that although generally sporozoite maturation starts after attachment of the moulted tick stage onto the host, T. equi sporozoites can mature prior to tick moulting.
Mark Walker states that pathogens e.g. Babesia are passed to the host in saliva while feeding. Feeding does not commence immediately on attachment, and typically a tick is attached for >12 hours before saliva is pumped into the host and blood removed.
We're struggling to get a definite answer. Until we do, we would suggest that it is likely that parasites do not pass from tick to horse until a tick has been attached for around 48 hours, but it is possible that they can pass before then, so check horses as frequently as possible for ticks during "tick seasons", and remove ticks as soon as they are found.
Jalovecka M, Hajdusek O, Sojka D, Kopacek P, Malandrin L
The Complexity of Piroplasms Life Cycles
Front. Cell. Infect. Microbiol. 23 July 2018 https://doi.org/10.3389/fcimb.2018.00248
Walker, MD
The biology and ecology of the Sheep Tick Ixodes ricinus
Antenna: The Royal Entomological Society, 2018, 42(2) 61-65
Q. How long must a tick be on a horse before piroplasmosis is transmitted?
A. This will depend on how quickly the tick's saliva passes into the horse, and how quickly the parasite sporozoites are released into the saliva.
The RESPE article Piroplasmoses suggests that transmission of the piroplasmosis parasites happens a few days after the tick becomes attached, because the parasites have to go through a development phase, and says that ticks should be removed as soon as they are seen. Sporozoites are the form of the parasite that pass from the tick to the horse, and they generally start to mature in the tick's salivary glands ready for transmission to the horse only once the tick attaches itself to the horse and starts to feed on blood. Once mature, sporozoites pass into the horse's blood in the tick's saliva as the tick feeds. However, in other Theileria species, sporozoites have been found to mature before the tick attaches for feeding, when temperatures are high, so it may be that T. equi can pass to a horse within hours of a tick attaching in hot weather (however the ticks in the CHG area of France tend to not be active in hot weather). Jalovecka 2018 states that although generally sporozoite maturation starts after attachment of the moulted tick stage onto the host, T. equi sporozoites can mature prior to tick moulting.
Mark Walker states that pathogens e.g. Babesia are passed to the host in saliva while feeding. Feeding does not commence immediately on attachment, and typically a tick is attached for >12 hours before saliva is pumped into the host and blood removed.
We're struggling to get a definite answer. Until we do, we would suggest that it is likely that parasites do not pass from tick to horse until a tick has been attached for around 48 hours, but it is possible that they can pass before then, so check horses as frequently as possible for ticks during "tick seasons", and remove ticks as soon as they are found.
Jalovecka M, Hajdusek O, Sojka D, Kopacek P, Malandrin L
The Complexity of Piroplasms Life Cycles
Front. Cell. Infect. Microbiol. 23 July 2018 https://doi.org/10.3389/fcimb.2018.00248
Walker, MD
The biology and ecology of the Sheep Tick Ixodes ricinus
Antenna: The Royal Entomological Society, 2018, 42(2) 61-65
Q. My vet has suggested a vitamin B12 injection to help restore the blood following the anaemia caused by the piroplasmosis - will this help?
A. Very unlikely. Horses, unlike humans, make B12 in their gut in association with cobalt, and have never been reported deficient in vitamin B12, and when B12 injections have been given to horses, the B12 was cleared rapidly from the blood, and excreted in the dung via bile fluid. Many good feed balancers (like Spillers Lite + Lean) will provide a small amount of B12 "just in case", and a forage diet will generally provide good levels of cobalt.
A. Very unlikely. Horses, unlike humans, make B12 in their gut in association with cobalt, and have never been reported deficient in vitamin B12, and when B12 injections have been given to horses, the B12 was cleared rapidly from the blood, and excreted in the dung via bile fluid. Many good feed balancers (like Spillers Lite + Lean) will provide a small amount of B12 "just in case", and a forage diet will generally provide good levels of cobalt.
Q. Would a liver, kidney or blood "detox" help a horse after having piroplasmosis?
A. Both severe piroplasmosis, and treatment with Imidocarb, can damage/affect the liver and kidneys. Raised liver (GGT, AST, ALP, LDH) and kidney (urea) enzymes may be seen during acute EP disease, and it has been suggested that this may be because of reduced blood flow/oxygen to the liver (presumably due to the hemolytic anaemia, so likely to be seen only in severe cases). Treating with Imidocarb to reduce parasite numbers is likely to be the best solution for liver or kidney damage due to the disease, plus correct diet to support recovery. When high doses of Imidocarb were given to horses to test the lethal dose (the LD50 - dose that killed half the horses - was 16 mg/kg), death was caused by liver and kidney damage. However, when normal doses of Imidocarb were given to ponies, no serious deleterious effects on liver function were seen, and blood GGT and bile acids were not raised, although transient increases in urinary GGT:creatinine ratios and mild azotemia have been seen, suggesting that transient changes in kidney function are possible during treatment. As part of the follow up after treating a horse for piroplasmosis, liver and kidney enzymes should be checked. If they are fine, don't worry. If they aren't, talk to your vet.
Just suppose the liver and/or kidneys were damaged - would a "detox" help? The short answer is no, you cannot detox the liver or kidneys (they ARE the body's detox organs). You may be able to help substances in the gastrointestinal (GI) system pass through, e.g. with activated carbon given on vet's advice if a horse has ingested poison, but nothing you can give will pull harmful substances out of the tissues/organs. Besides, what "toxins" are you talking about? The parasites? - the Imidocarb is dealing with them. What you should do is make sure that what goes into the horse isn't putting too much strain on either organ - so this would not be a good time to take a chance on feeding slightly mouldy hay or feed that might contain mycotoxins, or to feed huge amounts of low quality protein. Excess vitamin D has also been shown to damage the kidneys so don't exceed recommended levels in feed (3,00 IU/day for a 500 kg horse - NRC Nutrient Requirements of Horses 2007), although requirements for the fat soluble vitamins (A, D, E and K) should be met by the diet. Your horse will need protein, but it should be a quality protein which closely matches the horse's requirements for protein, so that there isn't too much waste for the liver and kidneys to process - quality proteins include soya and whey protein. Sugar beet and wheat bran with their high branched chain (leucine, isoleucine and valine) amino acid to aromatic (tyrosine, tryptophan, phenylalanine) amino acid ratios would be good feeds to reduce the accumulation of ammonia (source: Equine Applied and Clinical Nutrition 2013).
Could a "detox" product do harm? Yes, if it has an active ingredient that changes something in the body, and needs to be processed by the liver or kidneys, putting more strain on these organs. Some "detox" products have diuretic properties - the last thing you want to give a potentially dehydrated horse, or a horse with possible kidney damage. Some active ingredients have contra-indications with prescribed drugs, so be very careful about giving anything that hasn't been prescribed by your vet.
Ensure the horse has access to plenty of clean water, and is drinking well.
Detox: Save Your Money! Dr Eleanor Kellon March 2017
Four Words That I Dislike - David Ramey 2017
A. Both severe piroplasmosis, and treatment with Imidocarb, can damage/affect the liver and kidneys. Raised liver (GGT, AST, ALP, LDH) and kidney (urea) enzymes may be seen during acute EP disease, and it has been suggested that this may be because of reduced blood flow/oxygen to the liver (presumably due to the hemolytic anaemia, so likely to be seen only in severe cases). Treating with Imidocarb to reduce parasite numbers is likely to be the best solution for liver or kidney damage due to the disease, plus correct diet to support recovery. When high doses of Imidocarb were given to horses to test the lethal dose (the LD50 - dose that killed half the horses - was 16 mg/kg), death was caused by liver and kidney damage. However, when normal doses of Imidocarb were given to ponies, no serious deleterious effects on liver function were seen, and blood GGT and bile acids were not raised, although transient increases in urinary GGT:creatinine ratios and mild azotemia have been seen, suggesting that transient changes in kidney function are possible during treatment. As part of the follow up after treating a horse for piroplasmosis, liver and kidney enzymes should be checked. If they are fine, don't worry. If they aren't, talk to your vet.
Just suppose the liver and/or kidneys were damaged - would a "detox" help? The short answer is no, you cannot detox the liver or kidneys (they ARE the body's detox organs). You may be able to help substances in the gastrointestinal (GI) system pass through, e.g. with activated carbon given on vet's advice if a horse has ingested poison, but nothing you can give will pull harmful substances out of the tissues/organs. Besides, what "toxins" are you talking about? The parasites? - the Imidocarb is dealing with them. What you should do is make sure that what goes into the horse isn't putting too much strain on either organ - so this would not be a good time to take a chance on feeding slightly mouldy hay or feed that might contain mycotoxins, or to feed huge amounts of low quality protein. Excess vitamin D has also been shown to damage the kidneys so don't exceed recommended levels in feed (3,00 IU/day for a 500 kg horse - NRC Nutrient Requirements of Horses 2007), although requirements for the fat soluble vitamins (A, D, E and K) should be met by the diet. Your horse will need protein, but it should be a quality protein which closely matches the horse's requirements for protein, so that there isn't too much waste for the liver and kidneys to process - quality proteins include soya and whey protein. Sugar beet and wheat bran with their high branched chain (leucine, isoleucine and valine) amino acid to aromatic (tyrosine, tryptophan, phenylalanine) amino acid ratios would be good feeds to reduce the accumulation of ammonia (source: Equine Applied and Clinical Nutrition 2013).
Could a "detox" product do harm? Yes, if it has an active ingredient that changes something in the body, and needs to be processed by the liver or kidneys, putting more strain on these organs. Some "detox" products have diuretic properties - the last thing you want to give a potentially dehydrated horse, or a horse with possible kidney damage. Some active ingredients have contra-indications with prescribed drugs, so be very careful about giving anything that hasn't been prescribed by your vet.
Ensure the horse has access to plenty of clean water, and is drinking well.
Detox: Save Your Money! Dr Eleanor Kellon March 2017
Four Words That I Dislike - David Ramey 2017
Cases of Equine Piroplasmosis
Scoles GA, Hutcheson HJ, Schlater JL, Hennager SG, Pelzel AM, Knowles DP
Equine Piroplasmosis Associated with Amblyomma cajennense Ticks, Texas, USA
EID Oct 2011 Vol 17, No. 10
In October 2009 1 mare in Texas showed clinical signs of equine piroplasmosis and was found positive for T. equi by ELISA. 359 horses on the ranch were tested by ELISA, 292 (81%) were seropositive for T. equi. Ticks were found on 228 horses.
104 ticks (taken from horses that were 93% positive for T. equi) were placed on an uninfected horse 30 October. That horse had a fever (>39'C) 14 days after tick introduction, but no other clinical signs. Sighting of parasitized RBCs on blood slide peaked at 0.3% on day 17. Serology and PCR confirmed T. equi infection.
29 ticks (taken from horses that were at least 64% positive for T. equi) were placed on a second uninfected horse 30 October. That horse had a slight fever (39'C) 15 days after tick introduction, but no other clinical signs. Parasites were not seen on blood slides. PCR was positive for T. equi 42 days, and ELISA 87 days after tick introduction.
This research showed that T. equi can be transmitted interstadially, i.e. by a tick feeding on an infected horse and then infecting another horse, without going through a developmental change (however, the ticks were removed from the infected horses, they didn't drop off naturally). Note that raised temperature was the only clinical sign seen in both the intentionally infected horses.
Scoles GA, Hutcheson HJ, Schlater JL, Hennager SG, Pelzel AM, Knowles DP
Equine Piroplasmosis Associated with Amblyomma cajennense Ticks, Texas, USA
EID Oct 2011 Vol 17, No. 10
In October 2009 1 mare in Texas showed clinical signs of equine piroplasmosis and was found positive for T. equi by ELISA. 359 horses on the ranch were tested by ELISA, 292 (81%) were seropositive for T. equi. Ticks were found on 228 horses.
104 ticks (taken from horses that were 93% positive for T. equi) were placed on an uninfected horse 30 October. That horse had a fever (>39'C) 14 days after tick introduction, but no other clinical signs. Sighting of parasitized RBCs on blood slide peaked at 0.3% on day 17. Serology and PCR confirmed T. equi infection.
29 ticks (taken from horses that were at least 64% positive for T. equi) were placed on a second uninfected horse 30 October. That horse had a slight fever (39'C) 15 days after tick introduction, but no other clinical signs. Parasites were not seen on blood slides. PCR was positive for T. equi 42 days, and ELISA 87 days after tick introduction.
This research showed that T. equi can be transmitted interstadially, i.e. by a tick feeding on an infected horse and then infecting another horse, without going through a developmental change (however, the ticks were removed from the infected horses, they didn't drop off naturally). Note that raised temperature was the only clinical sign seen in both the intentionally infected horses.
More about ticks
3 species of tick that spread equine piroplasmosis are found in France:
Ixodidae species, particularly Ixodes ricinus or Castor bean tick/sheep tick - see below (also found in the UK)
Dermacentor reticulatus or Ornate cow tick - found outside of forests, active until winter (also found in the UK)
Rhipicephalus sanguineus or Brown dog tick - found mostly in the southern and Mediterranean area of France, and mostly on dogs (not found in the UK).
3 species of tick that spread equine piroplasmosis are found in France:
Ixodidae species, particularly Ixodes ricinus or Castor bean tick/sheep tick - see below (also found in the UK)
Dermacentor reticulatus or Ornate cow tick - found outside of forests, active until winter (also found in the UK)
Rhipicephalus sanguineus or Brown dog tick - found mostly in the southern and Mediterranean area of France, and mostly on dogs (not found in the UK).
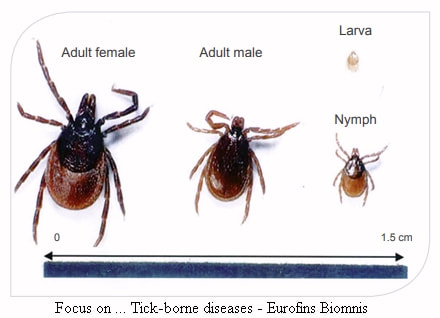
Ixodes ricinus is a hard tick (only hard ticks carry piroplasmosis) of the Ixodidae family, and commonly called the sheep tick or castor bean tick. It spreads piroplasmosis, Lyme disease and other tick-born diseases. A relatively small brown tick, males are 2.5-3 mm long and covered in a hard outer, females 3-4 mm long and only partially covered (female adults swell considerably (engorge) when eating, becoming over 10 mm long and light grey).
The sheep tick is the commonest tick in northern Europe, widespread in France and the UK, and is spreading north. It needs a moist, temperate climate with relative humidity of at least 80% where the litter layer and soil remain humid during the day, and lives in areas with good vegetation, particularly deciduous woodland, woodland edges and unkempt, particularly damp, grassland where there is a thick layer of vegetation on the ground to enable ticks to keep well hydrated, and where hosts are available. It is most active between 7 and 25'C, with numbers increasing in spring and autumn (depending on weather, habitat and host availability), and cold winters spent in the ground. Prolonged temperatures >34'C kill ticks, and temperatures below -10'C increase mortality.
The sheep tick is the commonest tick in northern Europe, widespread in France and the UK, and is spreading north. It needs a moist, temperate climate with relative humidity of at least 80% where the litter layer and soil remain humid during the day, and lives in areas with good vegetation, particularly deciduous woodland, woodland edges and unkempt, particularly damp, grassland where there is a thick layer of vegetation on the ground to enable ticks to keep well hydrated, and where hosts are available. It is most active between 7 and 25'C, with numbers increasing in spring and autumn (depending on weather, habitat and host availability), and cold winters spent in the ground. Prolonged temperatures >34'C kill ticks, and temperatures below -10'C increase mortality.
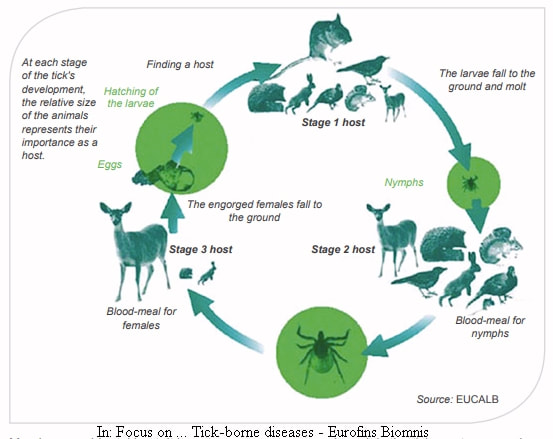
It is a 3 host tick, with each life stage feeding on a range of different & progressively larger hosts, including horses, cows, deer, boar and humans. The life cycle can take 3+ years and consists of egg, larva (0.8 mm long), nymph (1.4 mm long), adult. There must be a blood meal, which can last 3 to 10 day, on a single but different host before dropping into vegetation and moulting to the next stage. Adult males may not feed at all, they mate with the adult female as she engorges with blood on the host. Once mated and engorged, she drops off and lays up to 2,000 eggs in vegetation on the ground over 4 to 6 weeks, then dies. The larvae hatch after around 8 weeks, and do not move far from the nest. Sheep ticks "quest" for hosts - they climb to the tips of vegetation (grass, plants, bushes but not trees), hang on by their back legs, wait for a host to brush past, using sensors that detect heat, CO2 and vibrations, and attach to the host with their front legs. When not questing they live in the mat/leaf layer on the ground.
Once on the host the tick seeks a thin skinned area, cuts the skin and inserts its feeding tube, which it "glues" in place. Saliva containing anti-coagulants, pain killers and immune response inhibitors as well as disease pathogens is passed into the host; typically a tick is attached for more than 12 hours before saliva is pumped into the host and blood removed.
Once on the host the tick seeks a thin skinned area, cuts the skin and inserts its feeding tube, which it "glues" in place. Saliva containing anti-coagulants, pain killers and immune response inhibitors as well as disease pathogens is passed into the host; typically a tick is attached for more than 12 hours before saliva is pumped into the host and blood removed.
References:
Ixodes ricinus - Factsheet for experts - ECDC 2014
Focus on.. Tick-borne diseases - Eurofins Biomnis 2017
Rakover, Pauline PhD thesis November 2018
La Piroplasmose: Parasites, Vecteurs et Traitements
Ixodes ricinus - Factsheet for experts - ECDC 2014
Focus on.. Tick-borne diseases - Eurofins Biomnis 2017
Rakover, Pauline PhD thesis November 2018
La Piroplasmose: Parasites, Vecteurs et Traitements
Information is given as a guide only and must not be relied upon. Please consult your vet for all equine medical matters.